Day 1 :
Keynote Forum
Angeline Poulon Quintin
ICMCB, University of Bordeaux, France
Keynote: Nano structuration of thin films an alternative solution for hard chromium coating replacement
Time : 09:30-10:05

Biography:
Angeline Poulon-Quintin is an Associate Professor at the University of Bordeaux and ICMCB. She has a long experience in the correlation between process parameters, textures, microstructures and properties of structural and functional materials. Her current interests range from the search for innovative multifunctional coatings to the development of green processes to elaborate intermetallic compounds and/or nanostructured materials with innovative architecture, for applications in energy, aerospace and aeronautical industries. She is a Specialist in fine characterization with an extended recognized experience in electronic microscopy and physico-chemical techniques. She has co-authored 36 peer-reviewed articles, 40 oral presentations, 14 invited conferences and 4 patents.
To present & exhibit your MATERIALS @ our upcoming series PS: Materials Conferences | Materials Chemistry Conferences | Materials Chemistry 2020
Abstract:
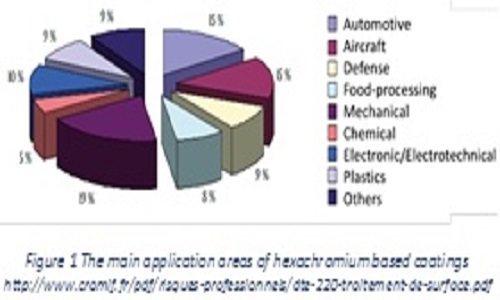
Hard chrome plating is used by Original Equipment Manufacturer to provide sealing surface for hydraulic seals, wear resistance to moving parts and corrosion protection to a wide variety of components for aircraft. The main areas of hexachromium using processes are mechanical, automotive, aerospace and military. By improving tribological properties (superior hardness) and corrosion resistance (natural ability to react with element such as oxygen), hard chromium can lower energy consumption of moving parts and machinery, reduce the need to replace parts frequently, leading to reduce waste and improve efficiency. The hard chrome plating is a powerful, simple and cheap process. From an environmental point of view, this process itself involves the use of highly toxic substances, such Cr6+ and lead compounds The Restriction of Hazardous substances Directive (Reach) was issue, banning several toxic substances including Cr6+. Hard chromium electroplating has been also classified by the U.S. Environmental Protection Agency (EPA) as an environmentally unfriendly process. Now, several hard chrome plating alternatives have been developed depending on the application:1) High Velocity Oxy-Fuel thermal sprays for repairs and for the deposition of high-quality hard-metals and metals coatings, 2) Electro and electroless plates; 3) Vacuum coating, 4) Heat treatments, 5) Laser and weld coatings. Due to its complex mix of properties, no single coating will replace hexavalent chromium in all applications. In this presentation, the exploration of the potential of nanostructured coating elaborated with innovative processes, will be discuss as an efficient alternative solution for hard chromium coating replacement. The opportunity to use architecture coating will be presented. Various families of materials will illustrate the correlation between process-Nano structuration and final properties.
Keynote Forum
Tsan Yao Chen
National Tsing Hua University, Taiwan
Keynote: Atomic scaled surface decoration promises oxygen reduction performance of nanocatalyst
Time : 11:35-12:10

Biography:
Tsan-Yao Chen is an Associate Professor in the Department of Engineering and System Science at National Tsing Hua University. With more than 10 years of experience in Materials Characterization and 6 years’ experience in electronic device (MEMS and IC) failure analysis of multiple executives by Synchrotron Light Source Techniques (at NSRRC, Taiwan). He conducts fundamental materials development in green energy applications including solar cell, fuel cell, and CO2 conversion, water sensing with worldwide collaboration at National rank research team in Russia, USA, Italy, Japan (SPring-8), and UK. He is also hosting research projects from academic and industrial funds on physical chemistry researches and environment sensing technologies.
To present & exhibit your MATERIALS @ our upcoming series PS: Materials Conferences | Materials Chemistry Conferences | Materials Chemistry 2020
Abstract:
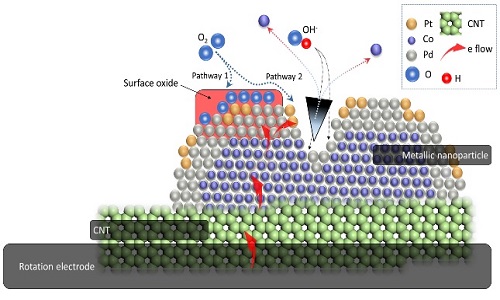
Surface decoration is an effective assessment for improving chemical stability of nanocatalysts. In this study, we demonstrate that the dimension of the surface decoration can be manipulated down to atomic scale. Apart from using noble metals, atomic scaled Pt clusters were employed to improve the oxygen reduction reaction performance of catalysts. Those clusters are decorated in surface defect regions of Cocore-Pdshell (Co-Pd) nanoparticles by using self-aligned nanocrystal growth followed by atomic quench with strong reduction agent. They localize electrons from neighboring atoms and boost activity of Co-Pd NP in ORR. With a proper reaction time and loading control, the Pt cluster decorated Co - Pd nanoparticles enhance its mass activity by 340 times as compared to that of commercial Pt catalysts in an alkaline electrolyte of 1.0M KOH.
Schematic representation for the atomic decoration in core-shell nanoparticle and the corresponding oxygen reduction reaction pathways.
- Materials Science and Chemistry | Materials Science and Engineering | Materials Chemistry in Developing Areas | Analytical Techniques and Instrumentation in Materials Chemistry | Polymeric Materials
Location: St. Gallen II

Chair
Debajyoti Ghoshal
Jadavpur University, India
Session Introduction
Thieo E Hogen Esch
University of Southern California, USA
Title: Nano-scale membranes based on polystyrene sulfonic acid blends
Time : 12:10-12:30

Biography:
Thieo E Hogen Esch has completed his PhD at the University of Leiden, Netherlands. Since then he has carried out research in the areas of physical organic chemistry and in the stereochemistry of anionic vinyl polymerization, the synthesis of block copolymers by anionic and free radical polymerizations at the University of Florida and since 1988 at the University of Southern California (USC). He has pioneered the synthesis and properties of “perfluorocarbon functionalized polymers” a topic that remains current. During the last few years his research has also included the synthesis and properties of thermodynamically unstable polymer blends of “camouflage mediated” homogeneous hydrophobic-hydrophilic type membrane.
To present & exhibit your MATERIALS @ our upcoming series PS: Materials Conferences | Materials Chemistry Conferences | Materials Chemistry 2020
Abstract:
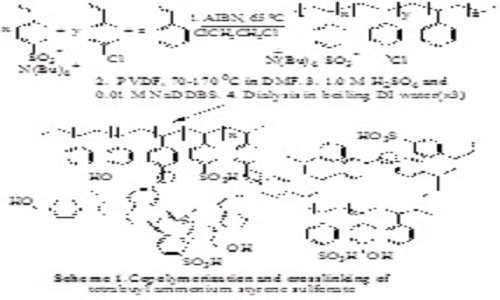
Flexible, strong, colorless and transparent polyvinylidenefluoride-olystyrenesulfonic acid (PVDF-PSSA) proton exchange membranes1-3 were synthesized by first mixing DMF solutions of PVDF and tetrabutylammonium polystyrenesulfonate (PTBASS) terpolymer having smaller (<20%) mole fractions of styrene (S) and 4-chloromethylstyrene (CMS). The tetrabutylammonium styrene (BASS) sulfonate precursor monomer was synthesized through simple ion exchange of sodium styrene sulfonate and tetrabutylammonium-chloride or bromide. The PBASS terpolymer precursor was synthesized through a conventional radical copolymerization of BASS, styrene and CMS. The PVDF-PBASS blends are formed by dissolution of the two polymers in DMF, followed by evaporation and thermal cross-linking through annealing at about 170°C. The EAS type crosslinking appears to involve the formation of TBA benzyl sulfonates that may directly benzylate the S and CMS units of the terpolymer. The PVDF-PBASS blends are optically transparent above about 165°C and remain so after rapid cooling followed by ion exchange mediated by a mixture of 1.0 M H2SO4 and surfactants and repeated aqueous dialysis giving low (<40 wt%) water content PVDF-PSSA membranes.4 Transmission electron microscopy confirms the presence of small (≥5 nm) PSSA domains the size and nature of which is still unknown. The water uptake, proton conductivity, ion exchange capacity (IEC), and methanol permeability of the membranes are tunable through variation of the PVDF/PSSA content and the sulfonic acid content of the terpolymer. At higher PSSA contents (>20 wt.%) the membranes were shown to have proton conductivities comparable or higher than Nafion-117 but significantly lower methanol permeabilities. These low cost, environmentally appealing and conveniently accessible membranes can be used in direct methanol fuel cells (DMFCs) and other applications.
Vincent Ball
University of Strasbourg, France
Title: From prebiotic chemistry to materials science: Deposition of aminomalononitrile based films
Time : 12:30-12:50

Biography:
Vincent Ball has studied Physics and Physical Chemistry at the Université Louis Pasteur (ULP), in Strasbourg, where he got his PhD in 1996. After a Postdoc at the Biozentrum Basel from 1996 to 1997, he was appointed as the Assistant Professor in Analytical Chemistry at ULP and Professor in Chemical Engineering in 2005. His research interests focus on thermodynamics and kinetics of self-assembly at Interfaces, biomimetic chemistry and the design of biomaterials with designed electrochemical and mechanical properties. He published a monography “Self-assembly processes at Interfaces-Multiscale phenomena” at Academic Press) in 2017. He was an Invited Researcher at the Michigan University in 2007 as a Fulbright fellow as well as in CSIRO-Clayton in 2018.
To present & exhibit your MATERIALS @ our upcoming series PS: Materials Conferences | Materials Chemistry Conferences | Materials Chemistry 2020
Abstract:
Surface functionalization with strongly adhesive and easy to functionalize films has remained highly materials specific for a long time. But bio inspiration from the mussel which is able to adhere to almost all known materials under wet conditions has allowed the development of polydopamine films from the oxidation of dopamine. Other molecules seem promising in this perspective: inspiration from prebiotic chemistry and from the famous experience realized by Prof. Miller which allowed it to demonstrate that strongly adhesive and biocompatible films can be deposited at solid-liquid interfaces from aminomalonitrile solutions. In this presentation it will be demonstrated that similar aminomalononitrile based films can be obtained by electropolymerization. The mechanism of the film deposition and their morphology (Figure) will also be described. In particular we will show that films can also be deposited at liquid-air interfaces but having morphology and a composition different from that deposited at solid-liquid interfaces.
A: water contact angles of AMN based films as a function of the deposition time, and B, C: film morphology after 1 and 19 h as characterized by SEM (B) and AFM (C).
Lunch Break 13:00-13:50 @ Foyer
Saikat Kumar Seth
Jadavpur University, India
Title: Salt-bridge (SB) interaction at work in building extended supramolecular networks: Experimental and theoretical study
Time : 13:50-14:10

Biography:
Saikat Kumar Seth is an Assistant Professor of Physics at Jadavpur University, Kolkata, India. He was awarded PhD at Indian Association for the Cultivation of Science (IACS), Kolkata in 2012 and Postdoctoral at Universitat de Illes Balears, Palma, SPAIN. His area of research is X-ray Crystallography and Crystal Engineering. The focus of his research is to explore weak noncovalent interactions in the context of Crystal Engineering along with high-performance computational studies. He had published 72-research articles in various peer-reviewed journals, edited one book (National level) and contributed a book chapter in Royal Society of Chemistry (RSC). He got various awards like Young Scientist, Best Poster, Best Presentation, and Research Excellence, Outstanding Reviewer award from 4 Elsevier Journals and Publons Peer Review Award in global peer review.
To present & exhibit your MATERIALS @ our upcoming series PS: Materials Conferences | Materials Chemistry Conferences | Materials Chemistry 2020
Abstract:
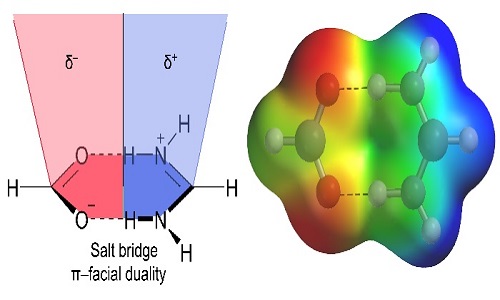
A series of pyridinium-carboxylate salts were designed prepared and structurally characterized to explore the importance of salt-bridge (SB) interactions in building self-assembled structures. We present a comprehensive analysis of the SB interaction in crystal structures of 4, 4′-Oxybis (benzoic acid) with substituted aminopyridines where the SBs displays extremely well defined geometric preferences. In the solid-state, compound exhibit lone-pair (l.p) ··· (SB)/(SB)···π+ assemblies while compound shows C‒H···(SB)/(SB)···π network. Interestingly, compound exhibit two distinct networks π+···(SB)/(SB)···(SB)/(SB)···π+ and C‒H···(SB)/(SB)···H‒C. These unexplored extended supramolecular networks are evident and explored for the first time in crystal structures. Our study also describes the duality of the salt bridge in establishing π-facial interactions with electron rich and/or electron poor moieties depending on the positive or negative nature of the SB counterpart that interacts with the SB surface. We describe the energetic and geometric features of salt-bridge interaction and explore its impact on the resultant supramolecular organization using high-level theoretical DFT-D3 calculations. The theoretical study combines the energetic features of the noncovalent forces that participate in the extended network and the characterization of the diverse interactions by means of Bader’s theory of ‘‘atoms in molecules’’.
Partha Pratim Ray
Jadavpur University, India
Title: Piezo response of defects mediated Methyl Ammonium Lead Iodide (MAPbI3)
Time : 14:10-14:30

Biography:
Partha Pratim Ray is working as an Associate Professor in the Department of Physics, at Jadavpur University, Kolkata, India. He has done his PhD and Postdoctoral research work on thin film silicon based solar cell. He is working on synthesis and application of different nanostructured materials in energy harvesting devices. He has studied the effect of different synthesis methods on charge transport properties which determines device performance. He is also working on metal organic framework based thin film photosensitive Schottky diode.
To present & exhibit your MATERIALS @ our upcoming series PS: Materials Conferences | Materials Chemistry Conferences | Materials Chemistry 2020
Abstract:
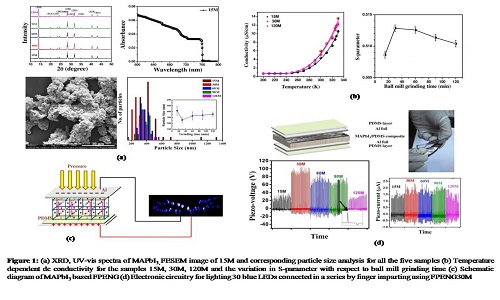
Formation of lattice defects (point defects/dislocations/grain boundaries) is an unavoidable phenomenon which is associated with almost all synthesis procedures following fast formation kinetics. Therefore a polycrystalline thin film of hybrid perovskite developed for a solar cell invariably contains lattice defects. Similarly in a systematic fashion using ball mill grinding technique we have synthesized perovskite material, Methylammonium lead iodide (MAPbI3) with different degrees of crystal defects as probed by positron annihilation spectroscopy (PAS) and utilized to fabricate flexible piezoelectric nanogenerators (FPENGs). Five sets of MAPbI3 samples are prepared by ball mill grinding procedure with different grinding time (15m, 30m, 60m, 90m and 120m). The formation and morphology of MAPbI3 is confirmed from their powder XRD pattern and field emission scanning electron microscopy (FESEM) images. The optical band gaps (1.63 eV) of all the samples are calculated from their absorption onset at 760 nm. The x-ray diffraction pattern suggests the formation of tetragonal crystal phase. We have demonstrated that at room temperature the lattice defects play the pivotal role in governing the ionic polarization from temperature dependent dc conductivity measurement and establish one-to-one correlation with the lattice defects as probed by PAS, which in principle governs the piezo-effect in MAPbI3. Here, we have shown that lattice defect mediated ionic polarization significantly changes VOC, but ISC remains almost same for all the samples as ISC has its origin on the value of piezoelectric constant and elastic modulus of the material. The best device performance is exhibited by maximum defect containing sample (30m) having significant amount of Pb2+ defects. A device fabricated with 5 wt % PDMS composite produces piezo-voltage (>100V) with a maximum power density of 0.3 mW/cm3 and can illuminate commercially available 30 blue light emitting diodes.
- Poster Presentations
Location: Foyer
Session Introduction
Sol M Mejia
Pontificia Universidad Javeriana, Colombia
Title: Selective adsorption of mercury (Hg2+) on a metal-organic framework obtained from aluminium and mercaptosuccinic acid

Biography:
Sol M Mejia is an Assistant Professor at the Chemistry Department of the Pontificia Universidad Javeriana in Colombia since 2014. She received her PhD from the Universidad de Antioquia in 2011. She did two postdoctoral researches, the first one at the University of Adelaide in Australia and the second one at the Universidad Autónoma Metropolitana in Mexico. She is the leader of the Computational Chemistry research line. Her major areas of interest are radical organic reactions, metal clusters, metal organic frameworks, and weak interactions (hydrogen bonds) as basic chemistry with possible applications in chemistry catalysis, biologic activity, new materials, and the like.
To present & exhibit your MATERIALS @ our upcoming series PS: Materials Conferences | Materials Chemistry Conferences | Materials Chemistry 2020
Abstract:
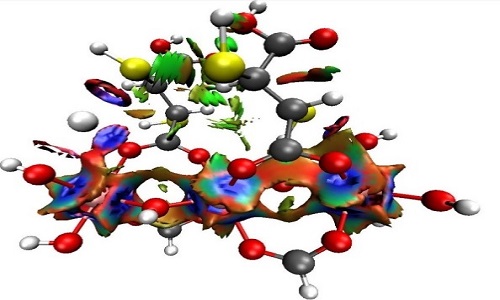
Water polluted with mercury represents a risk to the health of human beings and, in general, to all living beings that depend directly on these environments. It is then necessary to think about methods of capture and identification of chemicals species of mercury in water bodies. In this sense, the metalorganic frameworks (MOFs), hybrid crystalline materials, have a high potential to be used in the remediation of water contaminated with mercury. Through electronic structure calculations with density functional theory (DFT), the mechanisms of the interaction between Hg2+ and organic ligands were evaluated. These ligands were derived from fumaric acid when functionalized with groups -SH, - SCH3, -NH2, or -PH2. The interaction energy analysis, as well as the global reactivity indexes, suggest that the thiol group is the most selective to mercury compared to other water constituent metals such as calcium and lead (-56.40 kcal/mol, -48.99 kcal/mol, and -39.91 kcal/mol, respectively). This adsorptive capacity of the organic ligand was maintained by being part of a representative structure of the selected MOF. Through the analysis of charge transfer and non-covalent interactions index, it was verified that this adsorption was mediated by the tricoordination of mercury involving two oxygen atoms and the sulfur of the thiol group; these interactions are strong, mainly electrostatic, and not dispersive (see Figure 1). Taking as a basis the computational results, the Al-mercaptosuccinate MOF was synthesized and characterized. Its removal efficiency evidenced the high adsorptive capacity of this framework, reaching Hg2+ removal ratios of 99% at a concentration of the metal in aqueous solution of 10 μg/mL, in the same way this MOF shows to be selective against mercury in presence of other metals.
Isosurfaces obtained from the Non-Covalent Interactions (NCI) index at the MOF---Hg2+complex
Urs K Fischer
Ivoclar Vivadent AG, Liechtenstein
Title: Photoinitiating efficiency of visible light initiators based on tetraacyl derivatives of germanium and tin

Biography:
Urs K Fischer has his expertise in evaluation in research and development in the field of dental materials. His main topics are material characterization of new monomers, photoinitiators, and composites for dental applications.
To present & exhibit your MATERIALS @ our upcoming series PS: Materials Conferences | Materials Chemistry Conferences | Materials Chemistry 2020
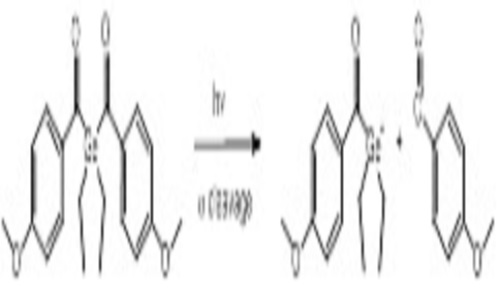
Abstract:
For several years, bisacylgermanium derivatives have been known to be very potent monomolecular initiators for photopolymerization induced by visible light (VL).[1] In particular, bis-(4-methoxybenzoyl) diethylgermane (Ivocerin®) exhibits an outstanding performance as amine-free Norrish type I photoinitiator (PI) for polymerization of dimethacrylate resins in dental materials and dimethacrylate-based composites, initialized by blue LED curing lights. Ivocerin® is characterized by strong absorption in the blue region of the VL spectrum, good solubility in and high PI reactivity towards dimethacrylate resins, good storage stability and excellent bleaching behavior. Recently, a very efficient synthetic method was developed, which allows the preparation of tetraacylgermanium derivatives according to a facile procedure in good yields. Based on the same synthetic procedure, the tin analogue tetrakis (2, 4, 6-trimethylbenzoyl) stannane is accessible, which displays a significant VL absorption beyond 500 nm.